Live-Cell Imaging
3. Real-Time Live-Cell
Imaging of Cellular Processes
Elevated pressure has been considered
the key risk factor for glaucoma [1,2]. Pressures of 100 mmHg
(acute glaucoma) or 30 mmHg (chronic glaucoma) cause apoptosis
of retinal ganglion cells (RGC's) and lead to the loss of
vision [3]. Although pressure induced apoptosis of RGC's has
been studied extensively through in-vivo [4,5,6] and in-vitro
[7,8] experiments, time evolution of RGC apoptosis had not
been investigated.
Here we present the first real-time
study of retinal ganglion cell apoptosis induced by elevated
pressure. This study utilizes differentiated RGC-5 cells as
the model system. A custom-built pressure chamber installed
on an inverted microscope allows us to image the apoptosis
process in real time. A few illustrative results of this study
are shown below.
1. Cell Morphology
Movie 1: Time-lapse phase-contrast images of differentiated
RGC-5 (retinal ganglion cell line) cells under 100mmHg pressure
for 20 hours
The cells in the movie illustrate the morphological symptoms
of apoptosis such as neurites retraction, cell body shrinkage,
and detachment from substrate.
2. Surface Apoptotic Marker:
Phosphatidyl Serine (PS)
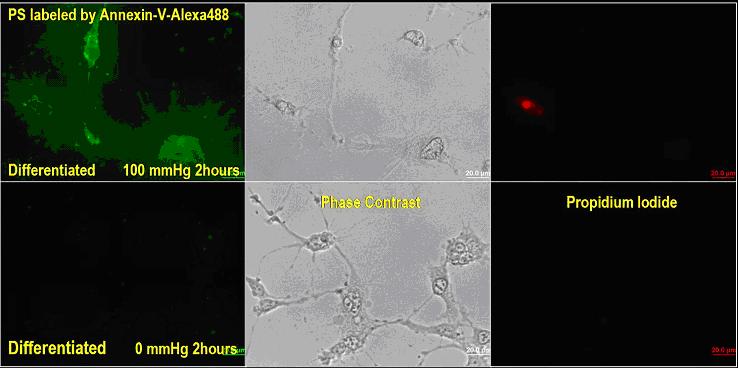
Phosphatidyl Serine (PS) is used
as an early-stage apoptosis marker since it is translocated
from inner bilipid membrane during normal status to outer
bilipid membrane during apoptosis. The translocated PS on
outer memebrane was detected with Alexa-488 fluorescent dye-conjugated
annexin-V. As shown in the image above (left column), higher
PS density was detected on the outer bilipid membrane of the
differentiated RGC-5 cells after 2 hours of exposure to 100
mmHg as compared to on the control cells with no exposure
to pressure. The lack of propidium iodide uptake in the cells
(right column) exposed to 100 mmHg for 2 hours demonstrates
that these cells were still in early apoptotic status, rather
than necrotic or late apoptotic status.
3. Cytosolic Apoptotic
Marker: Caspase-3 Activation
Movie 2: Time-lapse fluorescent images of activated caspase-3
in differentiated RGC-5 under 100mmHg pressure for 8 hours
Caspase-3 activation in these cells
was revealed by the increased fluorescent intensity from a
caspase-3 probe MR-DEVD. This study verifies the involvement
of Caspase cascade in the apoptosis of retinal ganglion cells.
Caspase-3 activation occurred ~6 hours after the application
of 100 mmHg pressure to the cells.
[1] N. OSBORNE, J. WOOD, G. CHIDLOW, J. BAE, J. MELENA, and
M. NASH, "Ganglion cell death in glaucoma: what do we
really know?", Br J Ophthalmol. 1999 August; 83(8): 980-986.
[2] Gülgün Tezel, "Oxidative Stress in Glaucomatous
Neurodegeneration: Mechanisms and Consequences", Prog
Retin Eye Res. 2006 September; 25(5): 490-513.
[3] Nickells RW, "From ocular hypertension to ganglion
cell death: a theoretical sequence of events leading to glaucoma",
Can J Ophthalmol. 2007 Apr;42(2):278-87.
[4] Tiande Shou, Jie Liu, Wei Wang, Yifeng Zhou, and Kanxing
Zhao, "Differential Dendritic Shrinkage of alpha and
beta Retinal Ganglion Cells in Cats with Chronic Glaucoma",
Invest Ophthalmol Vis Sci. 2003;44:3005-3010.
[5] Anderson DR, Hendrickson A, "Effect of intraocular
pressure on rapid axoplasmic transport in monkey optic nerve"
Invest Ophthalmol. 1974 Oct;13(10):771-783
[6] Quigley HA, McKinnon SJ, Zack DJ, Pease ME, Kerrigan-Baumrind
LA, Kerrigan DF, Mitchell RS, "Retrograde axonal transport
of BDNF in retinal ganglion cells is blocked by acute IOP
elevation in rats", Invest Ophthalmol Vis Sci. 2000 Oct;41(11):3460-6.
[7] Ju WK, Liu Q, Kim KY, Crowston JG, Lindsey JD, Agarwal
N, Ellisman MH, Perkins GA, Weinreb RN, "Elevated hydrostatic
pressure triggers mitochondrial fission and decreases cellular
ATP in differentiated RGC-5 cells", Invest Ophthalmol
Vis Sci. 2007 May;48(5):2145-51.
[8] Agar A, Li S, Agarwal N, Coroneo MT, Hill MA, "Retinal
ganglion cell line apoptosis induced by hydrostatic pressure",
Brain Res. 2006 May 1;1086(1):191-200.
Back to
Live-Cell Imaing Menu
|